- MID
- 3755758
Digital Olfactory Function Test Device
■ Reliable Data Accuracy: Our 32-level high-precision homogeneous fragrance dispersion technology delivers consistent fragrance concentration in every test, ensuring both accuracy and reproducibility of results.
■ Global Market Adaptability: The interchangeable fragrance cartridge system allows customization with scents familiar to different national and cultural backgrounds, enabling seamless global deployment.
■ Enhanced User-Friendly Experience: Provides voice guidance and visulaized scent images throught the entire test process, enhancing participant comprehension and elicit precise responses.
■ Ongoing Certification: Currently in progress for medical device regulatory approval.
- Certification
-
- Award
-
- Shipping
-
- Lead Time60 ~ 90days
- Type of Freight MT
- Modes of Transport FOB
- Products Shipped From Busan
- Payment
-
140+ Global Payments, trusted and secure payment provided by PayVerse.
Learn More
- Quantity
- MOQ : 10 EA
Detailed Description

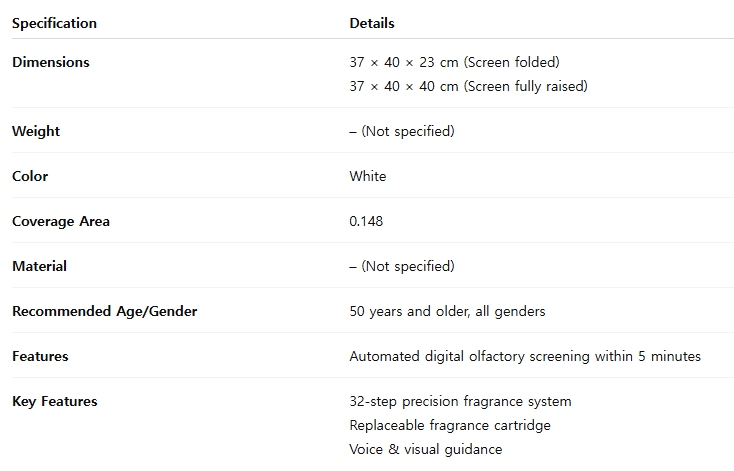
1. Product Features
- Rapid Screening: Enables screening for olfactory dysfunction, a key early indicator of dementia, within 5 minutes through an automated digital test.
- High-Precision Technology: A 32-step valve and pump control system dispenses a consistent and homogeneous scent for every test.
- Use of Familiar Scents: Equipped with replaceable cartridges, the device utilizes familiar, everyday scents tailored to different age groups. This approach minimizes test-related stress and enhances patient participation
- Compatibility with Cognitive Assessments: Can be used in conjunction with conventional interview-based methods, such as the MMSE, to improve the sensitivity and accuracy of the overall diagnosis.

- Scalability: Possesses the potential for expansion into other applications, including olfactory training and scent marketing platforms.
2. Detailed Description
The early detection of dementia has traditionally been constrained by high costs, patient fatigue, psychological resistance, and the limitations of subjective, questionnaire-based methods. In particular, conventional analog, pen-style olfactory tools rely on expensive imports, yield results that vary with the administrator's skill, and have limited applicability in Korea and other Asian regions due to their use of scents familiar only to Western cultures.
This product is a digital, automated olfactory testing device engineered to overcome these challenges. By applying a 32-step precision scent dispersion technology, it stably delivers an identical scent concentration for every test, allowing subjects to react intuitively to common and familiar scents.
Clinical validation has demonstrated a significant improvement in diagnostic performance. When the Mini-Mental State Examination (MMSE) was administered alone, the efficacy of early detection was limited to a sensitivity of 44% and a specificity of 89%. However, when combined with our olfactory-cognitive test, the performance improved to a sensitivity of 100% and a specificity of 84%.
Furthermore, based on our extensive experience in digital olfactory diagnostic clinical trials, a rapid pathway to commercialization as a medical device is highly feasible.
3. Benefits & Differentiators
- Transition from Analog to Digital: Shifts from manual, subjective methods to standardized, data-driven testing.
- Cultural Adaptability: Utilizes culturally appropriate scents for higher relevance and patient engagement.
- Quantitative Reliability: Ensures objective and reproducible results through precision engineering.
- Cost-Effectiveness: Reduces dependency on high-cost imported tools and optimizes clinical workflow.
- Integrated Diagnostic Support: Enhances the accuracy of existing cognitive assessments.
4. Considerations
- Periodic replacement of scent cartridges is required.
- Medical device certification is currently in progress.
5. Applications
- Medical Institutions: Dementia screening centers, hospitals, ENT (Ear, Nose, and Throat) clinics, and nursing homes.
- Public Health: Government-led or community-based early dementia detection programs.
- Extended Applications: Olfactory rehabilitation (training) and consumer-experience scent marketing platforms.











