- PID
- 3746762
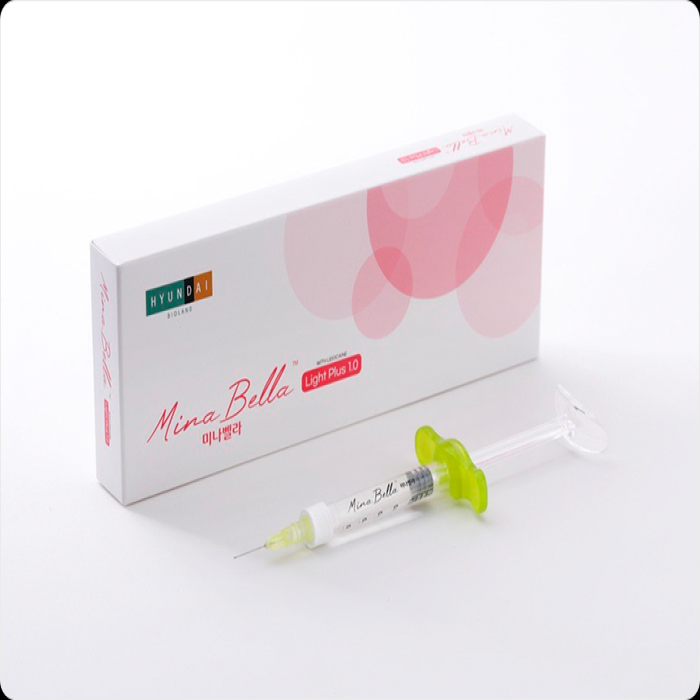
[Medical Device] MinaBella, HA Filler, HA Dermal Filler, KFDA Approval
MinaBella fillers are biomaterials for tissue reconstruction, used to achieve temporary improvement of adult nasolabial folds/wrinkles through physical reconstruction by subcutaneously injecting crosslinked hyaluronic acid containing lidocaine.
- The main ingredient of fillers Hyaluronic acid
Hyaluronic acid is the primary ingredient in MinaBella fillers, produced through a safe and clean process to ensure its safety for use. If you prefer, you can also use “active ingredient.”
- High-quality HA ensures production of high-quality and high-performance fillers.
- KFDA Approval
- Certification
-
- Award
-
- Shipping
-
- Payment
-
140+ Global Payments, trusted and secure payment provided by PayVerse.
Learn More
- Quantity
- MOQ : 10 Boxes
Product Summary
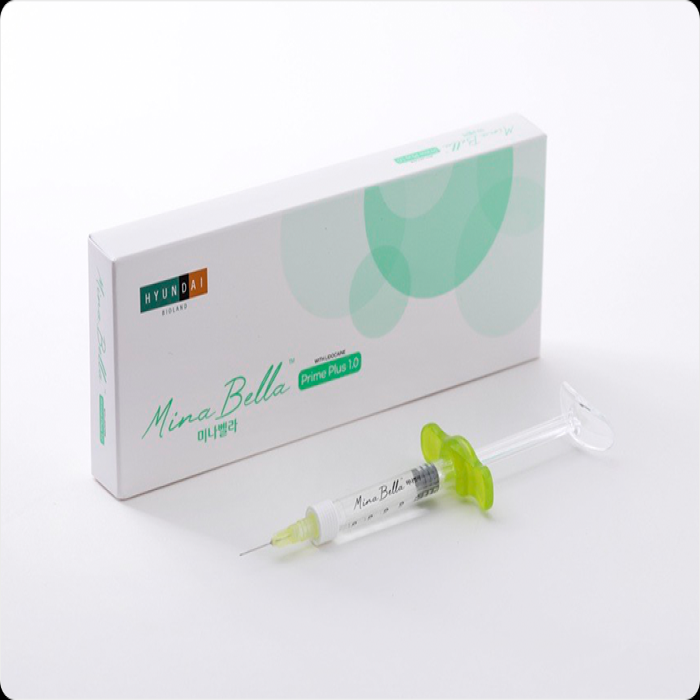
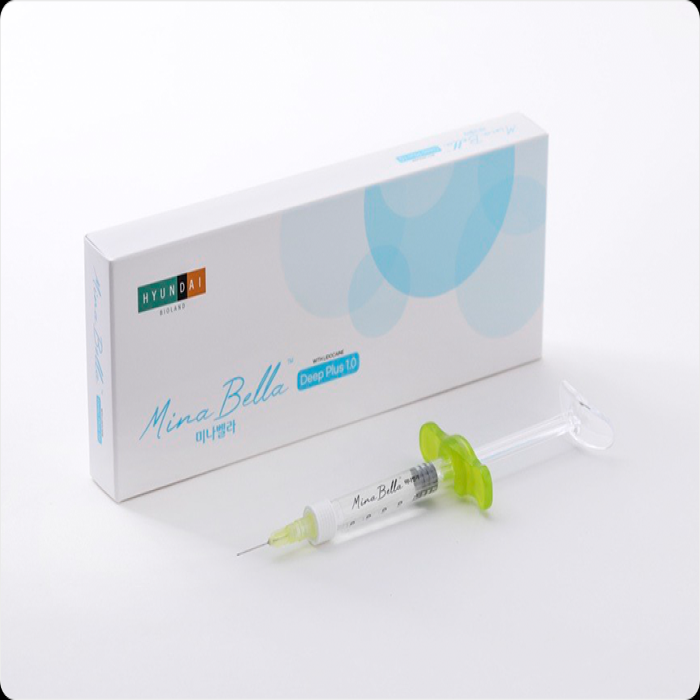
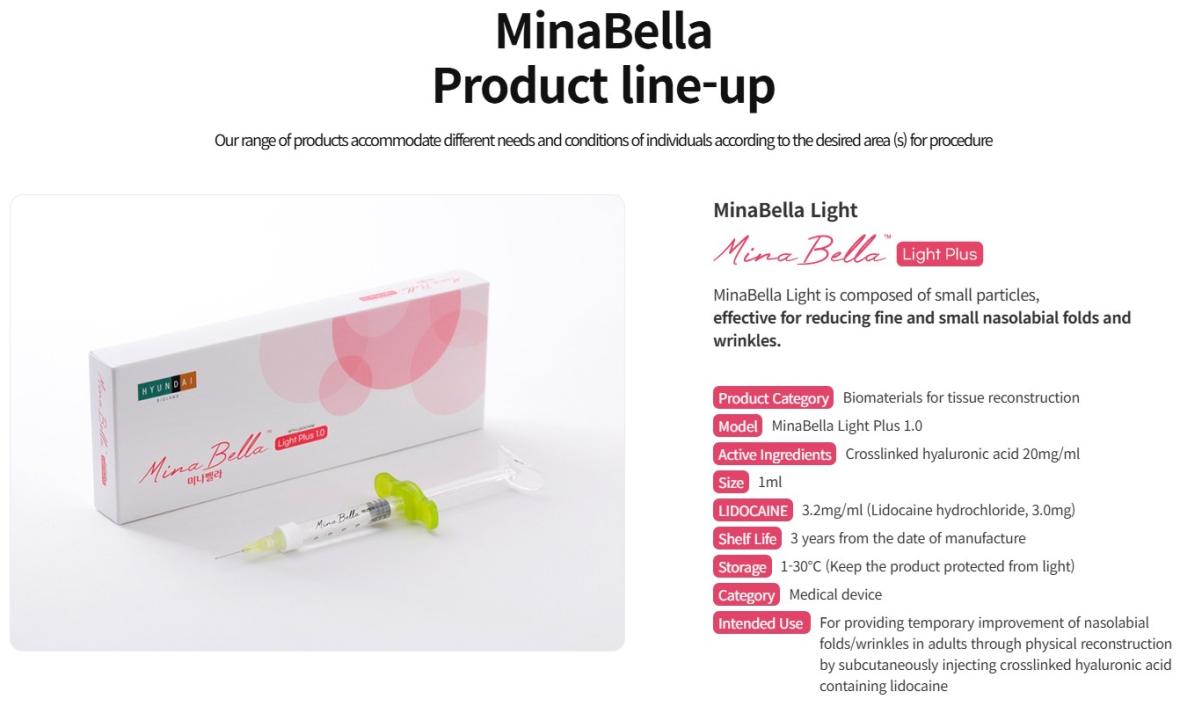
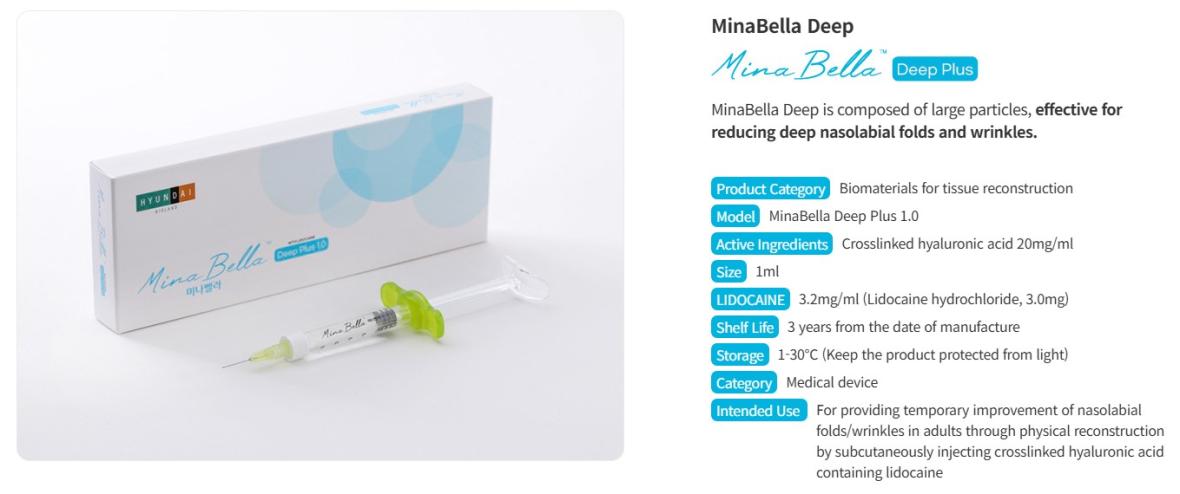
• Model : MinaBella Light Plus 1.0 / MinaBella Prime Plus 1.0 / MinaBella Deep Plus 1.0
• Active Ingredients : Crosslinked hyaluronic acid 20mg/ml
• Size : 1ml
• LIDOCAINE : 3.2mg/ml (Lidocaine hydrochloride, 3.0mg)
• Shelf Life : 3 years from the date of manufacture
• Storage : 1-30℃ (Keep the product protected from light)
• Category : Medical device
Detailed Description

Frequently Asked Questions
-
Where can I receive procedure with MinaBella fillers?
You can visit local dermatology or plastic surgery clinics or hospitals and undergo the procedure through consultation with healthcare professionals qualified as stipulated by appropriate local regulations
-
How much pain should I anticipate during the procedure?
Intensity of pain may vary depending on the site and the individual. MinaBella fillers contain lidocaine, a local anesthetic that may help alleviate pain during the procedure.
-
What are the precautions that I should keep in mind before and after the procedure?
Before the procedure, please refrain from wearing heavy makeup to ensure accurate treatment at the intended sites. Before the procedure, if you are taking any medications or have experienced a specific reaction to a specific medicine (or procedure), please consult with your doctor in advance. Since this product contains lidocaine, please inform your doctor (practitioner) in advance if you have any hypersensitivity to local anesthetics. Avoid drinking alcohol before and after the procedure. Within 12 hours after the procedure, avoid putting on any makeup. Avoid exposure to ultraviolet rays, UV gels, sauna, or steam rooms where possible for two weeks. Please refrain from cosmetic procedures such as laser treatment or facial peels for two weeks After the procedure, refrain from unnecessary, excessive touching of the site of procedure to allow the filler to settle.