- PID
- 3074847
FRABONE-I Bio-ceramics, Synthetic Bone Substitute
- Certification
-
- Award
-
- Shipping
-
- Modes of Transport EXW
- Payment
-
140+ Global Payments, trusted and secure payment provided by PayVerse.
Learn More
Detailed Description

[Company Introduction]
1. History
2010
• INOBONE Co. Ltd. was established
• Agreement with SoonChunHyang University (SCH) for co-research and development of bone substitutes
2011~2013
• INOBONE and SCH to co-develop bone substitutes
2014
• Successfully developed bone substitute FRABONE
• Obtained KFDA approval/permit for FRABONE
• Obtained KGMP approval/permit from KFDA
2015
• Obtained NET-99(New Excellent Technology) award from KMHW
• FRABONE launched in the market
• Obtained ISO 13485:2003 certification
2016
• Obtained NET-124(New Excellent Technology) award from KMHW
2017
Launched Frabone-I / Frabone-H
2018
Launched Frabone-W
2019
Obtained ISO 13485:2016 certification
2020
Obtained CE certification
2. Vision
Aims to become the leader of bone substitute developer in the global medical market
3. Business Model
1) Develops various innovative bone substitutes
2) Owns Core Technologies
- Nano bio-ceramic powders (HAp, BCP & TCP)
- Micro-structure control of Bio-ceramics
- Surface modification and high functionalization of Bio-ceramics
3) Human Bone Mimic Design Concept
FRABONE® comes in 0.7mm,1mm, 2mm and 3mm sizes to efficiently fill-up any shape and size of bone defects.
[Product Description]
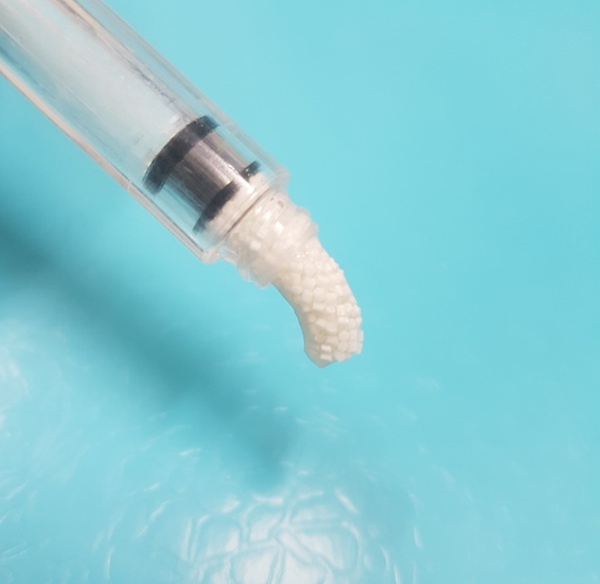
FRABONE is an innovative synthetic bone substitute that features micro-channels mimicking the strength and porosity of natural bones to provide faster bone regeneration.
[Product Features]
FRABONE®-I
FRABONE®-I has a unique design including 100 to 300 micropores that mimic the harversian canal structure of the human cortical bone which provides angiogenesis and nutrients through spaces between the granules and canals of FRABHONE®-Inject, which is essential for new bone growth. FRABONE®-I is innovative bone graft substitutes which overcome the inverse relationship between compressive strength and porosity of the existing bone graft materials. FRABONE®-I coated with hyaluronic acid has strong viscosity to enhance user's surgery convenience:
FRABONE®-I is a bone void filler intended for use as a synthetic bone implant material for orthopedic, dental, plastic surgery and neurosurgery applications as a filler for gaps and voids that are not intrinsic to the stability of the bony structure. FRABONE®-Inject is indicated to be packed gently into bony gaps or voids of the skeletal system, ie extremities, pelvis, femoral head, periodontal, oral, craniomaxillofacial and spine including use in posteriolateral spinal fusion procedures with appropriate stabilizing hardware. These defects may be osseous defects created surgically or from traumatic injury to the bone.
FRABONE®-I provides a bone void filler that resorbs and is replaced during the healing process.
Advangages of FRABONE and Particulars;
- Human Most similar to bone in composition, mechanical, strength and porosity
- Unique Design-mimicking Structure, The Haversian Canals
- Highly biocompatible and bioresorbable
- Tailored to bone grafting in the Apply Easily-service mark of Various-sized bone defects.
4. MOQ; 100 boxes
5. Product prices; To be discussed
6. Country of Origin; Korea
7. Product Summary; Solely for dental use such as cyst, GBR, Sinus lift
8. Production Capacity; We have production capacity enough to supply the demand.
For further detailed information, please visit our website: www.inobone.com